TAK-659 (hydrochloride)
TAK-659 (hydrochloride)
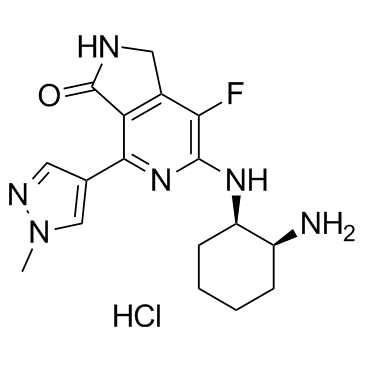
CAS: 1952251-28-3
Molecular Formula: C17H22ClFN6O
TAK-659 (hydrochloride) - Names and Identifiers
TAK-659 (hydrochloride) - Physico-chemical Properties
| Molecular Formula | C17H22ClFN6O |
| Molar Mass | 380.85 |
| Solubility | DMSO: 10 mM (Need Ultrasonic and Warming) |
| Storage Condition | -20℃ |
| In vitro study | In the cell proliferation assay, TAK-659 had an inhibitory effect on the SYK-dependent cell line (OCI-LY10). In B lymphocytes, the sensitivity of the cells to TAK-659 is associated with the mutations they carry that affect SYK activity. TAK-659 had no toxic effect on adherent primary tumor cells or solid tumor cell lines. The FLT3-ITD dependent cell lines (MV4-11 and MOLM-13) were sensitive to TAK-659 in a cell viability assay. RS4-11(ALL cell line) and RA1(Burkitt's lymphocyte line) with wild-type FLT3 were insensitive to TAK-659. In cultured human tumor cells, TAK-659 effectively inhibited the growth of cells of hematopoietic origin at the EC50 value (11-775 nM) that produced half the maximal response in the susceptible strain. Among many kinases, TAK-659 was more than 50-fold more selective for SYK and FLT3 than the other 290 proteins. In a co-culture system of primary CLL cells and Burkitt's lymphocytes, TAK-659 inhibited Syk activation and BCR signaling. In suspended primary CLL cell culture medium, TAK-659 caused a dose-dependent decrease in the phosphorylation of SykTyr525, Btk, NFκB, ERK1/2 and STAT3 after BCR stimulation. TAK-659 inhibition of Syk, induction of CLL cell apoptosis, inhibition of BCR and co culture source of survival signals. TAK-659 inhibition of chemotaxis to BMSC, CXCL12 and CXCL13, inhibition of microenvironment-induced chemoresistance in primary CLL cells. TAK-659 does not inhibit the molecular biological features of TCR signaling and T cell activation in primary T cells isolated from CLL patients. |
| In vivo study | In in vivo experiments in mice, TAK-659 prevented the expression of CD86 in peripheral blood B cells generated under anti-lgD stimulation. In a FLT3 dependent MV4-11 xenograft model, TAK-659 administration on 20 consecutive days at a dose of 60 mg/kg resulted in tumor regression. Preliminary pharmacokinetic data in plasma and urine indicate that TAK-659 is absorbed very quickly (average Tmax of 2-3 hours); At steady-state drug exposure, moderate variability (40-50% CV for DN-AUCtau); Mean peak-to-valley ratio of 3.2-4.2; Mean accumulation reached 2.1-2.6 fold after 15-day (once-daily) dosing. The renal clearance of the prototype drug accounts for 30-34% of the apparent oral clearance. TAK-659 administered via the oral route in patients with solid tumors or lymphoma, with considerable pharmacokinetic profile and safety, can be administered orally in continuous once-daily doses. |
TAK-659 (hydrochloride) - Preparation solution concentration reference
| 1mg | 5mg | 10mg | |
|---|---|---|---|
| 1 mM | 2.626 ml | 13.129 ml | 26.257 ml |
| 5 mM | 0.525 ml | 2.626 ml | 5.251 ml |
| 10 mM | 0.263 ml | 1.313 ml | 2.626 ml |
| 5 mM | 0.053 ml | 0.263 ml | 0.525 ml |
Last Update:2024-01-02 23:10:35
TAK-659 (hydrochloride) - Introduction
TAK-659 is a compound whose chemical name is TAK-659 hydrochloride. It is a solid powder, soluble in water.
TAK-659 (hydrochloride) is mainly used in the field of oncology research and drug development. It is a small molecule kinase inhibitor that can inhibit the activity of kinases in some signaling pathways, thereby interfering with the proliferation and survival of tumor cells. It is widely used in the laboratory to study the mechanism of cancer and to find new anti-cancer drug targets.
Regarding the production method, TAK-659 is produced by reacting TAK-659 with hydrochloric acid to produce TAK-659 hydrochloride.
In terms of safety, the use of TAK-659 (hydrochloride) should follow the relevant safety procedures. When used in the laboratory, wear appropriate protective equipment such as gloves and glasses to avoid contact with skin and eyes. In addition, it should be stored in a dry, cool place, away from fire and oxidizing agents.
However, the toxicity and side effects of TAK-659 may be related to its clinical use, so it should be fully studied and evaluated before use. At the same time, users should also follow the relevant regulations and guidelines to ensure its correct, safe and legal use.
TAK-659 (hydrochloride) is mainly used in the field of oncology research and drug development. It is a small molecule kinase inhibitor that can inhibit the activity of kinases in some signaling pathways, thereby interfering with the proliferation and survival of tumor cells. It is widely used in the laboratory to study the mechanism of cancer and to find new anti-cancer drug targets.
Regarding the production method, TAK-659 is produced by reacting TAK-659 with hydrochloric acid to produce TAK-659 hydrochloride.
In terms of safety, the use of TAK-659 (hydrochloride) should follow the relevant safety procedures. When used in the laboratory, wear appropriate protective equipment such as gloves and glasses to avoid contact with skin and eyes. In addition, it should be stored in a dry, cool place, away from fire and oxidizing agents.
However, the toxicity and side effects of TAK-659 may be related to its clinical use, so it should be fully studied and evaluated before use. At the same time, users should also follow the relevant regulations and guidelines to ensure its correct, safe and legal use.
Last Update:2024-04-09 21:49:45
Supplier List
Spot supply
Product Name: TAK-659 (hydrochloride) Visit Supplier Webpage Request for quotationCAS: 1952251-28-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: TAK-659 (hydrochloride) Request for quotation
CAS: 1952251-28-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 1952251-28-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: TAK-659 (hydrochloride) Request for quotation
CAS: 1952251-28-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1952251-28-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: TAK-659 (hydrochloride) Visit Supplier Webpage Request for quotation
CAS: 1952251-28-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1952251-28-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: TAK-659 (hydrochloride) Visit Supplier Webpage Request for quotationCAS: 1952251-28-3
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: TAK-659 (hydrochloride) Request for quotation
CAS: 1952251-28-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
CAS: 1952251-28-3
Tel: +86 13313090628
Email: 13313091926@163.com
Mobile: +86 13313090628
Wechat: 13373390591
WhatsApp: 18733928930
Product Name: TAK-659 (hydrochloride) Request for quotation
CAS: 1952251-28-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
CAS: 1952251-28-3
Tel: +86 19943533199
Email: vikki@api-made.com
QQ: 535948114
WhatsApp: +86 19943533199
Product Name: TAK-659 (hydrochloride) Visit Supplier Webpage Request for quotation
CAS: 1952251-28-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
CAS: 1952251-28-3
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History



